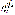返回
醋酸氯地孕酮
- 中文名称:
- 醋酸氯地孕酮
- 中文别名:
- 醋酸氯地孕酮;
氯化孕酮-17乙酸酯;
17α-Acetoxy-6-chloro-4,6-pregnadiene-3,20-dione;
氯地孕酮醋酸盐;
17-乙酰基-6-氯-6-脱氧孕酮
- 英文名称:
- Pregna-4,6-diene-3,20-dione,17-(acetyloxy)-6-chloro-
- 英文别名:
- Chlormadinone acetate;
6-chloro-17-hydroxypregna-4,6-diene-3,20-dione acetate;
Retex;
rs1280;
Lormin;
Natrol;
chlormadinone;
Verton;
17α-Acetoxy-6-chloro-4,6-pregnadiene-3,20-dione;
ST 155;
CMA;
Cero;
MATROL;
6-Chloro-17α-hydroxy-4,6-pregnadiene-3,20-dione 17-acetate,Gestafortin,Matrol;
6-Chloro-6-dehydro-17α-acetoxyprogesterone
- CAS No.:
- 302-22-7
- 分 子 式:
- C23H29ClO4
- 分 子 量:
- 404.93
- 精确分子量:
- 404.17500
- PSA:
- 60.44000
- MDL:
- MFCD00056471
- EINECS:
- 206-118-0
- BRN:
- 2633614
- InChI:
- InChI=1/C23H29ClO4/c1-13(25)23(28-14(2)26)10-7-18-16-12-20(24)19-11-15(27)5-8-21(19,3)17(16)6-9-22(18,23)4/h11-12,16-18H,5-10H2,1-4H3/t16?,17?,18?,21-,22+,23+/m1/s13
- 危险品标志:

- 风险术语:
- R60;R61;R40;R48
- 安全术语:
- S53;S22;S36/37/39;S45
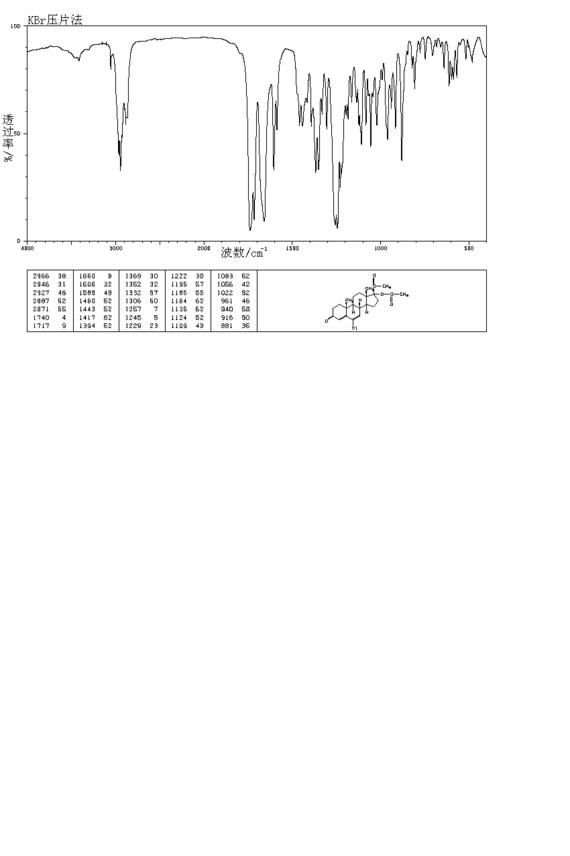
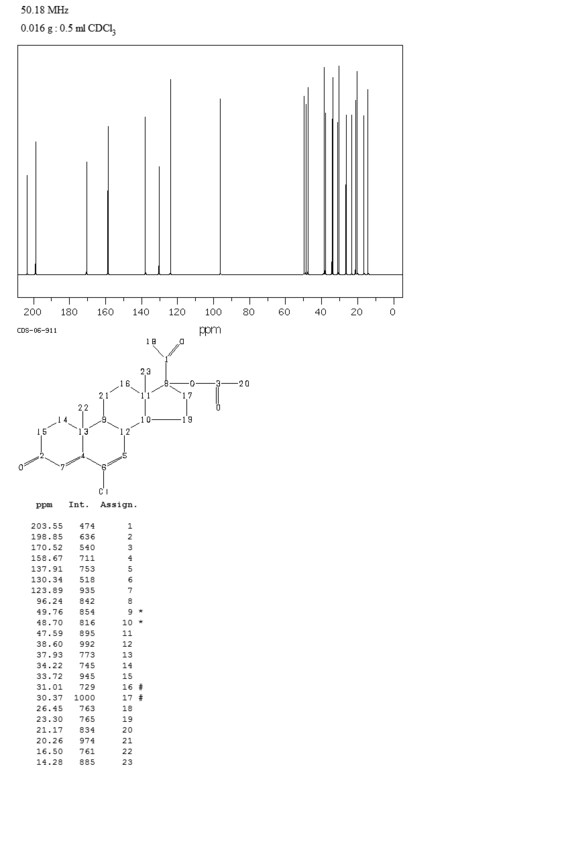
- 分子结构式:
-
- SDS:
- 查看
推荐供应商
上海源叶生物科技有限公司
氯地孕酮醋酸盐
- 联系人:Yuki
- 电 话:021-61845781
- 手 机:13585604150
- Cas号:302-22-7
- 包装:1g,5g
- 纯度:98%
- 价格:130.0元 - 400.0元
湖北鑫鸣泰化学有限公司
醋酸氯地孕酮
- 联系人:黄
- 电 话:027-50662354
- 手 机:13264726139
- Cas号:302-22-7
- 包装:100kg,200kg
- 纯度:99%
- 价格:1.0元 - 1.0元