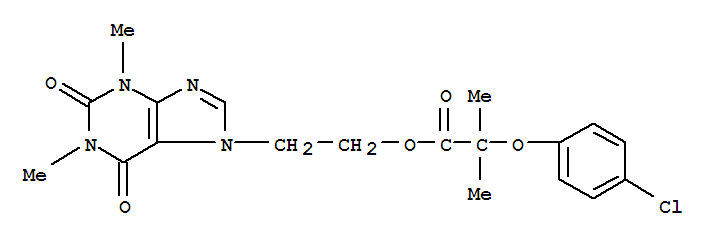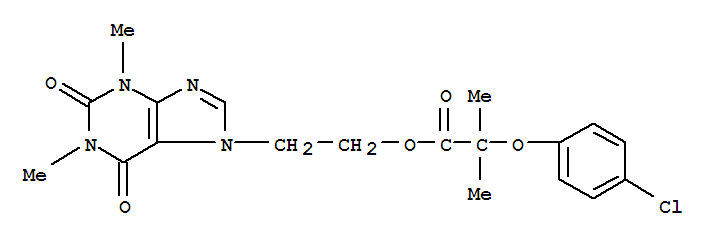益多酯
- 中文名称:
- 益多酯
- 中文别名:
- 益多酯;
乙羟茶碱安妥明酯
- 英文名称:
- Propanoic acid,2-(4-chlorophenoxy)-2-methyl-,2-(1,2,3,6-tetrahydro-1,3-dimethyl-2,6-dioxo-7H-purin-7-yl)ethyl ester
- 英文别名:
- 2-(1,3-dimethyl-2,6-dioxopurin-7-yl)ethyl 2-(4-chlorophenoxy)-2-methylpropanoate;
Etofyllinclofibrate;
Duolip;
Etofyllinclofibrat;
theofibrate;
Etofylline clofibrate
- CAS No.:
- 54504-70-0
- 分 子 式:
-
C19H21ClN4O5
- 分 子 量:
- 420.89
- 精确分子量:
- 420.12000
- PSA:
- 97.35000
- EINECS:
- 259-191-6
- InChI:
- The Key: KYAKGJDISSNVPZ-UHFFFAOYSA-N3
- 安全术语:
- Moderately toxic by ingestion and intraperitoneal routes. An experimental teratogen. Experimental reproductive effects. When heated to decomposition it emits very toxic fumes of Cl− and NOx. See also ESTERS.
- 分子结构式:
-
- SDS:
- 查看
益多酯 物化性质
- 密度:
- 1.37g/cm3
- 沸点:
- 615.8ºC at 760 mmHg
- 闪点:
- 326.2ºC
- 折射率:
- 1.617
益多酯 毒理性
CHEMICAL IDENTIFICATION
- RTECS NUMBER :
- UE9516500
- CHEMICAL NAME :
- Propionic acid, 2-(4-chlorophenoxy)-2-methyl-,
2-(1,2,3,6-tetrahydro-1,3-dimethyl-2,6-dioxo-
7H-purin-7-yl) ethyl ester
- CAS REGISTRY NUMBER :
- 54504-70-0
- LAST UPDATED :
- 199612
- DATA ITEMS CITED :
- 12
- MOLECULAR FORMULA :
- C19-H21-Cl-N4-O5
- MOLECULAR WEIGHT :
- 420.89
- WISWESSER LINE NOTATION :
- T56 BM DN FNVNVTJ D2OVX1&1&OR DG& F1 H1
HEALTH HAZARD DATA
ACUTE TOXICITY DATA
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 17000 mg/kg
- TOXIC EFFECTS :
- Liver - other changes
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 30,2023,1980
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 2830 mg/kg
- TOXIC EFFECTS :
- Liver - other changes
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 30,2023,1980
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Mammal - dog
- DOSE/DURATION :
- >10 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 27,1173,1977
** OTHER MULTIPLE DOSE TOXICITY DATA **
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 27300 mg/kg/26W-I
- TOXIC EFFECTS :
- Behavioral - fluid intake
Liver - changes in liver weight
Blood - changes in serum composition (e.g. TP, bilirubin, cholesterol)
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 30,2023,1980
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Mammal - pig
- DOSE/DURATION :
- 146 gm/kg/26W-I
- TOXIC EFFECTS :
- Liver - changes in liver weight
Blood - changes in serum composition (e.g. TP, bilirubin, cholesterol)
Nutritional and Gross Metabolic - weight loss or decreased weight gain
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 30,2023,1980
** REPRODUCTIVE DATA **
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 8250 mg/kg
- SEX/DURATION :
- female 5-15 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 30,2023,1980
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 4200 mg/kg
- SEX/DURATION :
- female 14 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - other measures of fertility
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 30,2023,1980
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 21 gm/kg
- SEX/DURATION :
- male 70 day(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Fertility - post-implantation mortality (e.g. dead and/or
resorbed implants per total number of implants)
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 30,2023,1980
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 7500 mg/kg
- SEX/DURATION :
- female 6-15 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 27,1173,1977
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 117 mg/kg
- SEX/DURATION :
- female 7-16 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - pre-implantation mortality (e.g. reduction in
number of implants per female; total number of implants per corpora lutea)
Reproductive - Effects on Embryo or Fetus - fetal death
Reproductive - Specific Developmental Abnormalities - other developmental
abnormalities
- REFERENCE :
- PHTOEH Pharmacology and Toxicology (Copenhagen). (Munksgaard International
Pub., POB 2148, DK-1016 Copenhagen K, Denmark) V.60- 1987-
Volume(issue)/page/year: 64,286,1989
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 5855 mg/kg
- SEX/DURATION :
- female 7-16 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
Reproductive - Specific Developmental Abnormalities - urogenital system
- REFERENCE :
- PHTOEH Pharmacology and Toxicology (Copenhagen). (Munksgaard International
Pub., POB 2148, DK-1016 Copenhagen K, Denmark) V.60- 1987-
Volume(issue)/page/year: 64,286,1989
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 7800 mg/kg
- SEX/DURATION :
- female 6-18 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - extra-embryonic structures
(e.g., placenta, umbilical cord)
Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 27,1173,1977
益多酯 分子结构与计算化学数据
计算化学数据
1.疏水参数计算参考值(XlogP):无
2.氢键供体数量:0
3.氢键受体数量:6
4.可旋转化学键数量:7
5.互变异构体数量:无
6.拓扑分子极性表面积:94
7.重原子数量:29
8.表面电荷:0
9.复杂度:653
10.同位素原子数量:0
11.确定原子立构中心数量:0
12.不确定原子立构中心数量:0
13.确定化学键立构中心数量:0
14.不确定化学键立构中心数量:0
15.共价键单元数量:1