吉美前列素
- 中文名称:
- 吉美前列素
- 中文别名:
- 吉美前列素;
吉美前列腺素;
Boc-Tyramine;
N-Boc-tyramine;
3,3,5,5-Tetramethylbenzidine.2HCl;
3,3',5,5'-Tetramethylbenzidine Dihydrochloride Monohydrate;
3,3',5,5'-Tetramethylbenzidine 2HCL;
4,4'-DIAMINO-3,3',5,5'-Tetramethylbiphenyl;
Dihydrochloride;
Dihydrochloride Monohydrate;
Dihydrochloride TMB;
TMB-HCL;
TMB DIHCL;
(TMB) Dihydrochloride;
3,3',5,5'-Tetramethylbiphenyl-4,4'-diamine dihydrochloride;
3,3,5,5-Tetramethylbenzidine HCL;
3,3',5,5'-Tetramethylbe
- 英文名称:
- Prosta-2,13-dien-1-oicacid, 11,15-dihydroxy-16,16-dimethyl-9-oxo-, methyl ester, (2E,11a,13E,15R)-
- 英文别名:
- gemeprost;
Cervagem;
Cervageme;
Cergem;
ONO 802;
Carprost;
Preglandin
- CAS No.:
- 64318-79-2
- 分 子 式:
-
C23H38O5
- 分 子 量:
- 394.54500
- 精确分子量:
- 394.27200
- PSA:
- 83.83000
- EINECS:
- 264-775-9
- InChI:
- InChI=1/C23H38O5/c1-5-6-15-23(2,3)21(26)14-13-18-17(19(24)16-20(18)25)11-9-7-8-10-12-22(27)28-4/h10,12-14,17-18,20-21,25-26H,5-9,11,15-16H2,1-4H3/b12-10+,14-13-/t17-,18-,20-,21+/m0/s13
- 分子结构式:
-
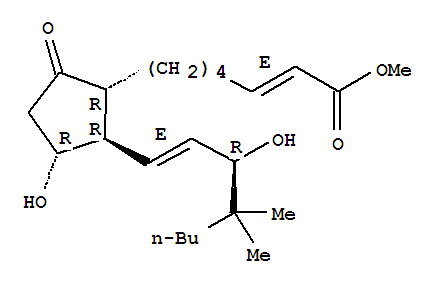
- SDS:
- 查看
吉美前列素 物化性质
- 密度:
- 1.08g/cm3
- 沸点:
- 510.5ºC at 760 mmHg
- 闪点:
- 163.9ºC
- 折射率:
- 1.533
- 存储条件/存储方法:
- 库房低温通风干燥
吉美前列素 毒理性
CHEMICAL IDENTIFICATION
- RTECS NUMBER :
- UK7894500
- CHEMICAL NAME :
- Prosta-2,13-dien-1-oic acid,
11,15-dihydroxy-16,16-dimethyl-9-oxo-, methyl ester,
(2E,11-alpha,13E,15R)-
- CAS REGISTRY NUMBER :
- 64318-79-2
- LAST UPDATED :
- 199709
- DATA ITEMS CITED :
- 25
- MOLECULAR FORMULA :
- C23-H38-O5
- MOLECULAR WEIGHT :
- 394.61
HEALTH HAZARD DATA
ACUTE TOXICITY DATA
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 56500 ug/kg
- TOXIC EFFECTS :
- Behavioral - somnolence (general depressed activity)
Lungs, Thorax, or Respiration - respiratory depression
Gastrointestinal - hypermotility, diarrhea
- REFERENCE :
- GEIRDK Gendai Iryo. Modern Medical Care. (Towa Bldg., 2-7 Nishiki-cho,
Kanda, Chiyoda-ku, Tokyo 101, Japan) V.1- 1969- Volume(issue)/page/year:
14,188,1982
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 22600 ug/kg
- TOXIC EFFECTS :
- Behavioral - altered sleep time (including change in righting reflex)
Lungs, Thorax, or Respiration - respiratory depression
- REFERENCE :
- GEIRDK Gendai Iryo. Modern Medical Care. (Towa Bldg., 2-7 Nishiki-cho,
Kanda, Chiyoda-ku, Tokyo 101, Japan) V.1- 1969- Volume(issue)/page/year:
14,188,1982
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 28600 ug/kg
- TOXIC EFFECTS :
- Behavioral - convulsions or effect on seizure threshold
Behavioral - ataxia
Lungs, Thorax, or Respiration - respiratory depression
- REFERENCE :
- GEIRDK Gendai Iryo. Modern Medical Care. (Towa Bldg., 2-7 Nishiki-cho,
Kanda, Chiyoda-ku, Tokyo 101, Japan) V.1- 1969- Volume(issue)/page/year:
14,188,1982
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravaginal
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 32500 ug/kg
- TOXIC EFFECTS :
- Gastrointestinal - hypermotility, diarrhea
- REFERENCE :
- GEIRDK Gendai Iryo. Modern Medical Care. (Towa Bldg., 2-7 Nishiki-cho,
Kanda, Chiyoda-ku, Tokyo 101, Japan) V.1- 1969- Volume(issue)/page/year:
14,188,1982
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 59 mg/kg
- TOXIC EFFECTS :
- Behavioral - somnolence (general depressed activity)
Lungs, Thorax, or Respiration - respiratory depression
Gastrointestinal - hypermotility, diarrhea
- REFERENCE :
- GEIRDK Gendai Iryo. Modern Medical Care. (Towa Bldg., 2-7 Nishiki-cho,
Kanda, Chiyoda-ku, Tokyo 101, Japan) V.1- 1969- Volume(issue)/page/year:
14,188,1982
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 32500 ug/kg
- TOXIC EFFECTS :
- Behavioral - altered sleep time (including change in righting reflex)
Lungs, Thorax, or Respiration - respiratory depression
- REFERENCE :
- GEIRDK Gendai Iryo. Modern Medical Care. (Towa Bldg., 2-7 Nishiki-cho,
Kanda, Chiyoda-ku, Tokyo 101, Japan) V.1- 1969- Volume(issue)/page/year:
14,188,1982
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 29500 ug/kg
- TOXIC EFFECTS :
- Behavioral - convulsions or effect on seizure threshold
Behavioral - ataxia
Lungs, Thorax, or Respiration - respiratory depression
- REFERENCE :
- GEIRDK Gendai Iryo. Modern Medical Care. (Towa Bldg., 2-7 Nishiki-cho,
Kanda, Chiyoda-ku, Tokyo 101, Japan) V.1- 1969- Volume(issue)/page/year:
14,188,1982
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravaginal
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 36 mg/kg
- TOXIC EFFECTS :
- Gastrointestinal - hypermotility, diarrhea
- REFERENCE :
- GEIRDK Gendai Iryo. Modern Medical Care. (Towa Bldg., 2-7 Nishiki-cho,
Kanda, Chiyoda-ku, Tokyo 101, Japan) V.1- 1969- Volume(issue)/page/year:
14,188,1982
** OTHER MULTIPLE DOSE TOXICITY DATA **
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 187500 ug/kg/30D-C
- TOXIC EFFECTS :
- Gastrointestinal - other changes
Liver - changes in liver weight
Blood - changes in leukocyte (WBC) count
- REFERENCE :
- GEIRDK Gendai Iryo. Modern Medical Care. (Towa Bldg., 2-7 Nishiki-cho,
Kanda, Chiyoda-ku, Tokyo 101, Japan) V.1- 1969- Volume(issue)/page/year:
14,188,1982
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 7500 ug/kg/30D-C
- TOXIC EFFECTS :
- Kidney, Ureter, Bladder - other changes in urine composition
Endocrine - changes in spleen weight
Blood - normocytic anemia
- REFERENCE :
- GEIRDK Gendai Iryo. Modern Medical Care. (Towa Bldg., 2-7 Nishiki-cho,
Kanda, Chiyoda-ku, Tokyo 101, Japan) V.1- 1969- Volume(issue)/page/year:
14,188,1982
** REPRODUCTIVE DATA **
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravaginal
- DOSE :
- 86 ug/kg
- SEX/DURATION :
- female 14 week(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - abortion
- REFERENCE :
- BJOGAS British Journal of Obstetrics and Gynaecology. (Blackwell Scientific
Pub. Ltd., POB 88, Oxford, UK) V.82- 1975- Volume(issue)/page/year:
91,1136,1984
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravaginal
- DOSE :
- 20 ug/kg
- SEX/DURATION :
- female 31 week(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - abortion
- REFERENCE :
- BJOGAS British Journal of Obstetrics and Gynaecology. (Blackwell Scientific
Pub. Ltd., POB 88, Oxford, UK) V.82- 1975- Volume(issue)/page/year:
96,745,1989
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravaginal
- DOSE :
- 20 ug/kg
- SEX/DURATION :
- female 10 week(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - abortion
- REFERENCE :
- CCPTAY Contraception. (Geron-X, Inc., POB 1108, Los Altos, CA 94022) V.1-
1970- Volume(issue)/page/year: 27,51,1983
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravaginal
- DOSE :
- 20 ug/kg
- SEX/DURATION :
- female 10 week(s) after conception
- TOXIC EFFECTS :
- Reproductive - Maternal Effects - uterus, cervix, vagina
- REFERENCE :
- CCPTAY Contraception. (Geron-X, Inc., POB 1108, Los Altos, CA 94022) V.1-
1970- Volume(issue)/page/year: 27,339,1983
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intraperitoneal
- DOSE :
- 4 mg/kg
- SEX/DURATION :
- female 2 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - pre-implantation mortality (e.g. reduction in
number of implants per female; total number of implants per corpora lutea)
- REFERENCE :
- NYKZAU Nippon Yakurigaku Zasshi. Japanese Journal of Pharmacology. (Nippon
Yakuri Gakkai, c/o Kyoto Daigaku Igakubu Yakurigaku Kyoshitsu, Konoe-cho,
Yoshida, Sakyo-ku, Kyoto 606, Japan) V.40- 1944- Volume(issue)/page/year:
79,15,1982
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intraperitoneal
- DOSE :
- 4 mg/kg
- SEX/DURATION :
- female 17 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - abortion
- REFERENCE :
- NYKZAU Nippon Yakurigaku Zasshi. Japanese Journal of Pharmacology. (Nippon
Yakuri Gakkai, c/o Kyoto Daigaku Igakubu Yakurigaku Kyoshitsu, Konoe-cho,
Yoshida, Sakyo-ku, Kyoto 606, Japan) V.40- 1944- Volume(issue)/page/year:
79,15,1982
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 2 mg/kg
- SEX/DURATION :
- female 1 day(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Fertility - other measures of fertility
- REFERENCE :
- NYKZAU Nippon Yakurigaku Zasshi. Japanese Journal of Pharmacology. (Nippon
Yakuri Gakkai, c/o Kyoto Daigaku Igakubu Yakurigaku Kyoshitsu, Konoe-cho,
Yoshida, Sakyo-ku, Kyoto 606, Japan) V.40- 1944- Volume(issue)/page/year:
79,15,1982
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravaginal
- DOSE :
- 80 ug/kg
- SEX/DURATION :
- female 17-20 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Newborn - viability index (e.g., # alive at day 4
per # born alive)
Reproductive - Effects on Newborn - weaning or lactation index (e.g., #
alive at weaning per # alive at day 4)
- REFERENCE :
- GEIRDK Gendai Iryo. Modern Medical Care. (Towa Bldg., 2-7 Nishiki-cho,
Kanda, Chiyoda-ku, Tokyo 101, Japan) V.1- 1969- Volume(issue)/page/year:
14,593,1982
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravaginal
- DOSE :
- 438 ug/kg
- SEX/DURATION :
- female 1-7 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - pre-implantation mortality (e.g. reduction in
number of implants per female; total number of implants per corpora lutea)
Reproductive - Fertility - litter size (e.g. # fetuses per litter; measured
before birth)
Reproductive - Effects on Embryo or Fetus - fetal death
- REFERENCE :
- GEIRDK Gendai Iryo. Modern Medical Care. (Towa Bldg., 2-7 Nishiki-cho,
Kanda, Chiyoda-ku, Tokyo 101, Japan) V.1- 1969- Volume(issue)/page/year:
14,809,1982
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravaginal
- DOSE :
- 438 ug/kg
- SEX/DURATION :
- female 1-7 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - extra-embryonic structures
(e.g., placenta, umbilical cord)
- REFERENCE :
- GEIRDK Gendai Iryo. Modern Medical Care. (Towa Bldg., 2-7 Nishiki-cho,
Kanda, Chiyoda-ku, Tokyo 101, Japan) V.1- 1969- Volume(issue)/page/year:
14,809,1982
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravaginal
- DOSE :
- 5 mg/kg
- SEX/DURATION :
- female 6-15 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
- REFERENCE :
- TCMUD8 Teratogenesis, Carcinogenesis, and Mutagenesis. (Alan R. Liss, Inc.,
41 E. 11th St., New York, NY 10003) V.1- 1980- Volume(issue)/page/year:
4,233,1984
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravaginal
- DOSE :
- 2 mg/kg
- SEX/DURATION :
- female 17-20 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - musculoskeletal system
Reproductive - Effects on Newborn - stillbirth
Reproductive - Effects on Newborn - live birth index (measured after birth)
- REFERENCE :
- GEIRDK Gendai Iryo. Modern Medical Care. (Towa Bldg., 2-7 Nishiki-cho,
Kanda, Chiyoda-ku, Tokyo 101, Japan) V.1- 1969- Volume(issue)/page/year:
14,593,1982
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intraperitoneal
- DOSE :
- 4 mg/kg
- SEX/DURATION :
- female 6-7 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - pre-implantation mortality (e.g. reduction in
number of implants per female; total number of implants per corpora lutea)
- REFERENCE :
- NYKZAU Nippon Yakurigaku Zasshi. Japanese Journal of Pharmacology. (Nippon
Yakuri Gakkai, c/o Kyoto Daigaku Igakubu Yakurigaku Kyoshitsu, Konoe-cho,
Yoshida, Sakyo-ku, Kyoto 606, Japan) V.40- 1944- Volume(issue)/page/year:
79,15,1982
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intraperitoneal
- DOSE :
- 200 ug/kg
- SEX/DURATION :
- female 24-25 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - abortion
- REFERENCE :
- NYKZAU Nippon Yakurigaku Zasshi. Japanese Journal of Pharmacology. (Nippon
Yakuri Gakkai, c/o Kyoto Daigaku Igakubu Yakurigaku Kyoshitsu, Konoe-cho,
Yoshida, Sakyo-ku, Kyoto 606, Japan) V.40- 1944- Volume(issue)/page/year:
79,15,1982
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravaginal
- DOSE :
- 162 ug/kg
- SEX/DURATION :
- female 6-18 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - post-implantation mortality (e.g. dead and/or
resorbed implants per total number of implants)
Reproductive - Fertility - abortion
- REFERENCE :
- TCMUD8 Teratogenesis, Carcinogenesis, and Mutagenesis. (Alan R. Liss, Inc.,
41 E. 11th St., New York, NY 10003) V.1- 1980- Volume(issue)/page/year:
4,225,1984
推荐供应商
郑州力本生物技术有限公司
吉美前列素
- 联系人:刘林
- 电 话:0371-86536307
- 手 机:13949119001
- Cas号:64318-79-2
- 包装:1-5/桶kg
- 纯度:99
- 价格:洽谈
台州市科德化工有限公司
吉美前列素
- 联系人:牟慧
- 电 话:0576-84613060
- 手 机:13093829633
- Cas号:64318-79-2
- 包装:洽谈
- 纯度:
- 价格:洽谈

