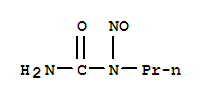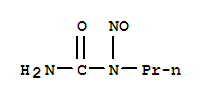丙基亚硝基脲
- 中文名称:
- 丙基亚硝基脲
- 中文别名:
- 1-nitroso-1-propylurea
- 英文名称:
- Urea,N-nitroso-N-propyl-
- 英文别名:
- 1-nitroso-1-propylurea;
UREA,N-NITROSO-N-PROPYL;
1-Propyl-1-nitrosourea;
N-Propylnitrosourea;
Nitrosopropylurea;
N-Nitroso-N-propylurea;
Urea,1-nitroso-1-propyl;
N-Nitrosopropylurea;
Propylnitrosourea;
1-nitroso-1-propyl-urea;
N-Propyl-N-nitrosourea
- CAS No.:
- 816-57-9
- 分 子 式:
-
C4H9N3O2
- 分 子 量:
- 131.16
- 精确分子量:
- 131.06900
- PSA:
- 75.76000
- InChI:
- The Key: YGGSSUISOYLCBF-UHFFFAOYSA-N3
- 安全术语:
- Suspected carcinogen with experimental carcinogenic, neoplastigenic, tumorigenic data. An experimental teratogen. Mutation data reported. When heated to decomposition it emits toxic fumes of NOx. See also N-NITROSO COMPOUNDS.
- 分子结构式:
-
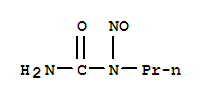
- SDS:
- 查看
丙基亚硝基脲 物化性质
- 密度:
- 1.27g/cm3
- 沸点:
- 200.3ºC at 760 mmHg
- 闪点:
- 74.9ºC
- 折射率:
- 1.524
丙基亚硝基脲 毒理性
CHEMICAL IDENTIFICATION
- RTECS NUMBER :
- YT9740000
- CHEMICAL NAME :
- Urea, N-nitroso-N-propyl-
- CAS REGISTRY NUMBER :
- 816-57-9
- LAST UPDATED :
- 199709
- DATA ITEMS CITED :
- 34
- MOLECULAR FORMULA :
- C4-H9-N3-O2
- MOLECULAR WEIGHT :
- 131.16
- WISWESSER LINE NOTATION :
- ZVN3&NO
HEALTH HAZARD DATA
ACUTE TOXICITY DATA
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 480 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 1375 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 1190 mg/kg/5D-I
- TOXIC EFFECTS :
- Related to Chronic Data - death
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 120 mg/kg
female 19 day(s) after conception
- TOXIC EFFECTS :
- Tumorigenic - equivocal tumorigenic agent by RTECS criteria
Reproductive - Tumorigenic effects - transplacental tumorigenesis
Brain and Coverings - tumors
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 200 mg/kg
- TOXIC EFFECTS :
- Tumorigenic - neoplastic by RTECS criteria
Reproductive - Tumorigenic effects - ovarian tumors
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 400 mg/kg
- TOXIC EFFECTS :
- Tumorigenic - equivocal tumorigenic agent by RTECS criteria
Blood - leukemia
Skin and Appendages - tumors
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Administration onto the skin
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 525 mg/kg/50W-I
- TOXIC EFFECTS :
- Tumorigenic - Carcinogenic by RTECS criteria
Skin and Appendages - tumors
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - hamster
- DOSE/DURATION :
- 3024 mg/kg/36W-C
- TOXIC EFFECTS :
- Tumorigenic - Carcinogenic by RTECS criteria
Vascular - tumors
Blood - tumors
- TYPE OF TEST :
- TD - Toxic dose (other than lowest)
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 2080 mg/kg/1Y-I
- TOXIC EFFECTS :
- Tumorigenic - equivocal tumorigenic agent by RTECS criteria
Gastrointestinal - tumors
Skin and Appendages - tumors
- TYPE OF TEST :
- TD - Toxic dose (other than lowest)
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - hamster
- DOSE/DURATION :
- 7728 mg/kg/23W-C
- TOXIC EFFECTS :
- Tumorigenic - Carcinogenic by RTECS criteria
Vascular - tumors
Blood - tumors
- TYPE OF TEST :
- TD - Toxic dose (other than lowest)
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 689 mg/kg/25W-C
- TOXIC EFFECTS :
- Tumorigenic - equivocal tumorigenic agent by RTECS criteria
Gastrointestinal - colon tumors
Endocrine - thyroid tumors
- TYPE OF TEST :
- TD - Toxic dose (other than lowest)
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 1054 mg/kg/30W-C
- TOXIC EFFECTS :
- Tumorigenic - equivocal tumorigenic agent by RTECS criteria
Gastrointestinal - colon tumors
Endocrine - thyroid tumors
- TYPE OF TEST :
- TD - Toxic dose (other than lowest)
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 420 mg/kg
- TOXIC EFFECTS :
- Tumorigenic - equivocal tumorigenic agent by RTECS criteria
Peripheral Nerve and Sensation - structural change in nerve or sheath
Skin and Appendages - tumors
- TYPE OF TEST :
- TD - Toxic dose (other than lowest)
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 500 mg/kg
- TOXIC EFFECTS :
- Tumorigenic - equivocal tumorigenic agent by RTECS criteria
Cardiac - tumors
Gastrointestinal - tumors
- TYPE OF TEST :
- TD - Toxic dose (other than lowest)
- ROUTE OF EXPOSURE :
- Administration onto the skin
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 525 mg/kg/50W-I
- TOXIC EFFECTS :
- Tumorigenic - equivocal tumorigenic agent by RTECS criteria
Skin and Appendages - tumors
- TYPE OF TEST :
- TD - Toxic dose (other than lowest)
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 10920 mg/kg/39W-C
- TOXIC EFFECTS :
- Tumorigenic - equivocal tumorigenic agent by RTECS criteria
Blood - lymphoma, including Hodgkin's disease
- TYPE OF TEST :
- TD - Toxic dose (other than lowest)
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 1969 mg/kg/35W-I
- TOXIC EFFECTS :
- Tumorigenic - equivocal tumorigenic agent by RTECS criteria
Gastrointestinal - tumors
Blood - leukemia
- TYPE OF TEST :
- TD - Toxic dose (other than lowest)
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 3 gm/kg/10W-C
- TOXIC EFFECTS :
- Tumorigenic - neoplastic by RTECS criteria
Gastrointestinal - tumors
Blood - lymphoma, including Hodgkin's disease
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Parenteral
- DOSE :
- 80 mg/kg
- SEX/DURATION :
- female 9 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetal death
Reproductive - Specific Developmental Abnormalities - Central Nervous System
Reproductive - Specific Developmental Abnormalities - eye/ear
- TYPE OF TEST :
- Micronucleus test
- TYPE OF TEST :
- Micronucleus test
- TYPE OF TEST :
- Specific locus test
MUTATION DATA
- TYPE OF TEST :
- DNA damage
- TEST SYSTEM :
- Mammal - species unspecified Lymphocyte
- DOSE/DURATION :
- 500 umol/L
- REFERENCE :
- CRNGDP Carcinogenesis (London). (Oxford Univ. Press, Pinkhill House,
Southfield Road, Eynsham, Oxford OX8 1JJ, UK) V.1- 1980-
Volume(issue)/page/year: 4,1455,1983