夫拉扎勃; 去脂舒; 呋咱甲氢龙
- 中文名称:
- 夫拉扎勃; 去脂舒; 呋咱甲氢龙
- 中文别名:
- 夫拉扎勃;
去脂舒
- 英文名称:
- Androstano[2,3-c][1,2,5]oxadiazol-17-ol,17-methyl-, (5a,17b)-
- 英文别名:
- Furazabol;
EINECS 214-983-0;
Myotolon;
Frazalon;
androfurazanol;
DH 245;
Miotolon
- CAS No.:
- 1239-29-8
- 分 子 式:
-
C20H30N2O2
- 分 子 量:
- 330.46
- 精确分子量:
- 330.23100
- PSA:
- 59.15000
- InChI:
- The Key: RGLLOUBXMOGLDQ-IVEVATEUSA-N3
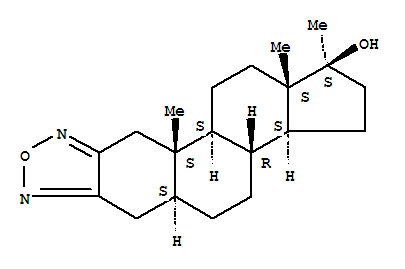
- 分子结构式:
-
- SDS:
- 查看
夫拉扎勃; 去脂舒; 呋咱甲氢龙 物化性质
- 密度:
- 1.145g/cm3
- 熔点:
- 152.5ºC
- 沸点:
- 445.6ºC at 760mmHg
- 闪点:
- 223.3ºC
- 折射率:
- 1.549
- 蒸汽压:
- 1.01E-08mmHg at 25°C
- 存储条件/存储方法:
- 库房通风低温干燥
夫拉扎勃; 去脂舒; 呋咱甲氢龙 毒理性
CHEMICAL IDENTIFICATION
- RTECS NUMBER :
- BV8075000
- CHEMICAL NAME :
- 5-alpha-Androstano(2,3-c)(1,2,5)oxadiazol-17-beta-ol,
17-methyl-
- CAS REGISTRY NUMBER :
- 1239-29-8
- LAST UPDATED :
- 199512
- DATA ITEMS CITED :
- 15
- MOLECULAR FORMULA :
- C20-H30-N2-O2
- MOLECULAR WEIGHT :
- 330.52
- WISWESSER LINE NOTATION :
- T G5 E6 B665 HNON&TTTTJ M1 Q1 RQ R1
HEALTH HAZARD DATA
ACUTE TOXICITY DATA
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- >4 gm/kg
- TOXIC EFFECTS :
- Lungs, Thorax, or Respiration - acute pulmonary edema
Gastrointestinal - ulceration or bleeding from duodenum
- REFERENCE :
- KSRNAM Kiso to Rinsho. Clinical Report. (Yubunsha Co., Ltd., 1-5, Kanda
Suda-Cho, Chiyoda-ku, KS Bldg., Tokyo 101, Japan) V.1- 1960-
Volume(issue)/page/year: 4,2031,1970
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 745 mg/kg
- TOXIC EFFECTS :
- Behavioral - altered sleep time (including change in righting reflex)
Behavioral - convulsions or effect on seizure threshold
- REFERENCE :
- KSRNAM Kiso to Rinsho. Clinical Report. (Yubunsha Co., Ltd., 1-5, Kanda
Suda-Cho, Chiyoda-ku, KS Bldg., Tokyo 101, Japan) V.1- 1960-
Volume(issue)/page/year: 4,2031,1970
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- >4 gm/kg
- TOXIC EFFECTS :
- Lungs, Thorax, or Respiration - acute pulmonary edema
Gastrointestinal - ulceration or bleeding from duodenum
- REFERENCE :
- KSRNAM Kiso to Rinsho. Clinical Report. (Yubunsha Co., Ltd., 1-5, Kanda
Suda-Cho, Chiyoda-ku, KS Bldg., Tokyo 101, Japan) V.1- 1960-
Volume(issue)/page/year: 4,2031,1970
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 1731 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 3,187,1969
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 494 mg/kg
- TOXIC EFFECTS :
- Behavioral - altered sleep time (including change in righting reflex)
Behavioral - convulsions or effect on seizure threshold
Behavioral - ataxia
- REFERENCE :
- CPBTAL Chemical and Pharmaceutical Bulletin. (Japan Pub. Trading Co., USA,
1255 Howard St., San Francisco, CA 94103) V.6- 1958-
Volume(issue)/page/year: 14,285,1966
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- >4 gm/kg
- TOXIC EFFECTS :
- Behavioral - altered sleep time (including change in righting reflex)
Behavioral - convulsions or effect on seizure threshold
Gastrointestinal - changes in structure or function of salivary glands
- REFERENCE :
- CPBTAL Chemical and Pharmaceutical Bulletin. (Japan Pub. Trading Co., USA,
1255 Howard St., San Francisco, CA 94103) V.6- 1958-
Volume(issue)/page/year: 14,285,1966
** REPRODUCTIVE DATA **
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 6 gm/kg
- SEX/DURATION :
- female 13-18 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetal death
Reproductive - Effects on Newborn - weaning or lactation index (e.g., #
alive at weaning per # alive at day 4)
- REFERENCE :
- KSRNAM Kiso to Rinsho. Clinical Report. (Yubunsha Co., Ltd., 1-5, Kanda
Suda-Cho, Chiyoda-ku, KS Bldg., Tokyo 101, Japan) V.1- 1960-
Volume(issue)/page/year: 4,2088,1970
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 4800 ug/kg
- SEX/DURATION :
- female 13-18 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - musculoskeletal system
Reproductive - Specific Developmental Abnormalities - urogenital system
- REFERENCE :
- KSRNAM Kiso to Rinsho. Clinical Report. (Yubunsha Co., Ltd., 1-5, Kanda
Suda-Cho, Chiyoda-ku, KS Bldg., Tokyo 101, Japan) V.1- 1960-
Volume(issue)/page/year: 4,2088,1970
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 1200 mg/kg
- SEX/DURATION :
- female 9-14 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - litter size (e.g. # fetuses per litter; measured
before birth)
Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- KSRNAM Kiso to Rinsho. Clinical Report. (Yubunsha Co., Ltd., 1-5, Kanda
Suda-Cho, Chiyoda-ku, KS Bldg., Tokyo 101, Japan) V.1- 1960-
Volume(issue)/page/year: 4,2088,1970
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 750 mg/kg
- SEX/DURATION :
- female 9-14 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetal death
- REFERENCE :
- KSRNAM Kiso to Rinsho. Clinical Report. (Yubunsha Co., Ltd., 1-5, Kanda
Suda-Cho, Chiyoda-ku, KS Bldg., Tokyo 101, Japan) V.1- 1960-
Volume(issue)/page/year: 4,2088,1970
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 30 mg/kg
- SEX/DURATION :
- female 9-14 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - urogenital system
- REFERENCE :
- KSRNAM Kiso to Rinsho. Clinical Report. (Yubunsha Co., Ltd., 1-5, Kanda
Suda-Cho, Chiyoda-ku, KS Bldg., Tokyo 101, Japan) V.1- 1960-
Volume(issue)/page/year: 4,2088,1970
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 1200 ug/kg
- SEX/DURATION :
- female 12 day(s) pre-mating
- TOXIC EFFECTS :
- Reproductive - Maternal Effects - menstrual cycle changes or disorders
- REFERENCE :
- CPBTAL Chemical and Pharmaceutical Bulletin. (Japan Pub. Trading Co., USA,
1255 Howard St., San Francisco, CA 94103) V.6- 1958-
Volume(issue)/page/year: 14,285,1966
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 2100 mg/kg
- SEX/DURATION :
- female 7-12 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
Reproductive - Specific Developmental Abnormalities - urogenital system
Reproductive - Effects on Newborn - stillbirth
- REFERENCE :
- KSRNAM Kiso to Rinsho. Clinical Report. (Yubunsha Co., Ltd., 1-5, Kanda
Suda-Cho, Chiyoda-ku, KS Bldg., Tokyo 101, Japan) V.1- 1960-
Volume(issue)/page/year: 4,2088,1970
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 2100 mg/kg
- SEX/DURATION :
- female 7-12 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Newborn - live birth index (measured after birth)
Reproductive - Effects on Newborn - weaning or lactation index (e.g., #
alive at weaning per # alive at day 4)
- REFERENCE :
- KSRNAM Kiso to Rinsho. Clinical Report. (Yubunsha Co., Ltd., 1-5, Kanda
Suda-Cho, Chiyoda-ku, KS Bldg., Tokyo 101, Japan) V.1- 1960-
Volume(issue)/page/year: 4,2088,1970
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- DOSE :
- 30 mg/kg
- SEX/DURATION :
- female 7-12 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- KSRNAM Kiso to Rinsho. Clinical Report. (Yubunsha Co., Ltd., 1-5, Kanda
Suda-Cho, Chiyoda-ku, KS Bldg., Tokyo 101, Japan) V.1- 1960-
Volume(issue)/page/year: 4,2088,1970
夫拉扎勃; 去脂舒; 呋咱甲氢龙 分子结构与计算化学数据
计算化学数据
1.疏水参数计算参考值(XlogP):4
2.氢键供体数量:1
3.氢键受体数量:4
4.可旋转化学键数量:0
5.互变异构体数量:无
6.拓扑分子极性表面积:59.2
7.重原子数量:24
8.表面电荷:0
9.复杂度:542
10.同位素原子数量:0
11.确定原子立构中心数量:7
12.不确定原子立构中心数量:0
13.确定化学键立构中心数量:0
14.不确定化学键立构中心数量:0
15.共价键单元数量:1

