返回
2-(2-呋喃基)-3-(5-硝基-2-呋喃基)丙烯酰胺
- 中文名称:
- 2-(2-呋喃基)-3-(5-硝基-2-呋喃基)丙烯酰胺
- 中文别名:
- 2-(2-呋喃基)-3-(5-硝基-2-呋喃基)丙烯酰胺;
2-(2-呋喃)-3-(5-硝基-2-呋喃)丙烯酰胺
- 英文名称:
- 2-Furanacetamide, a-[(5-nitro-2-furanyl)methylene]-
- 英文别名:
- (Z)-2-(2-furyl)-3-(5-nitro-2-furyl)acrylamide;
2--3-<5-nitro-furyl-(2)>-acrylsaeure-amid;
2-(2-Furyl)-3-(5-nitro-2-furyl)acrylamide;
(Z)-2-(furan-2-yl)-3-(5-nitrofuran-2-yl)prop-2-enamide;
Tofuron;
2-furan-2-yl-3-(5-nitro-furan-2-yl)-acrylamide;
FURYLFURAMIDE;
AF-2;
cis-Furylfuramide;
AF 2 (Preservative);
cis-AF 2;
Furylamide
- CAS No.:
- 3688-53-7
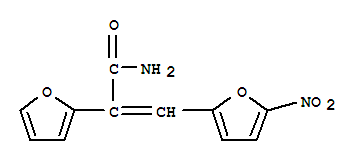
- 分 子 式:
- C11H8N2O5
- 分 子 量:
- 248.19
- 精确分子量:
- 248.04300
- PSA:
- 115.19000
- InChI:
- The Key: LYAHJFZLDZDIOH-UHFFFAOYSA-N3
- 分子结构式:
-
- SDS:
- 查看
推荐供应商
湖北恩星生物科技有限公司
2-(2-呋喃基)-3-(5-硝基-2-呋喃基)丙烯酰胺
- 联系人:邵经理
- 电 话:16621771607
- 手 机:16621771607
- Cas号:3688-53-7
- 包装:100kg,200kg,300kg,400kg,500kg
- 纯度:
- 价格:64.0元 - 100.0元
