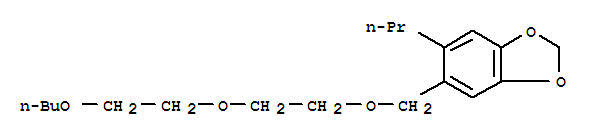返回
增效醚
- 中文名称:
- 增效醚
- 中文别名:
- 增效醚;
3,4-亚甲二氧基-6-正丙基苄基正丁基二缩乙二醇醚;
胡椒基丁醚;
5-(2-(2-丁氧基乙氧基)乙氧甲基)-6-丙基-1,3-苯并二氧杂戊环
- 英文名称:
- Piperonyl butoxide
- 英文别名:
- piperonyl butoxide;
5-{[2-(2-butoxyethoxy)ethoxy]methyl}-6-propyl-2H-1,3-benzodioxole;
INCITE;
5-[2-(2-butoxyethoxy)ethoxymethyl]-6-propyl-1,3-benzodioxole;
5-[[2-(2-butoxyethoxy)ethoxy]methyl]-6-propyl-1,3-benzodioxole;
PBO;
nia5273;
Piperonylbutoxide;
5-((2-(2-Butoxyethoxy)ethoxy)methyl)-6-propylbenzo[d][1,3]dioxole;
2-(2-butoxyethoxy)ethyl 6-propylpiperonyl ether;
Butocide;
BUTACIDE;
2-(2-Butoxyethoxy)ethyl (6-propylpiperonyl) ether;
Pyrenon;
Butacid;
PRENTOX;
Vex;
Piperonyl butoxide;
fmc5273;
5-[2-(2-Butoxyethoxy)ethoxymethyl]-6-propyl-1,3-benzodioxole;
piperonyl-butoxide
- CAS No.:
- 51-03-6
- 分 子 式:
-
C19H30O5
- 分 子 量:
- 338.44
- 精确分子量:
- 338.20900
- PSA:
- 46.15000
- MDL:
- MFCD00005842
- EINECS:
- 200-076-7
- BRN:
- 288063
- InChI:
- InChI=1/C15H22O3.C4H10O3/c1-3-5-7-16-10-13-9-15-14(17-11-18-15)8-12(13)6-4-2;5-1-3-7-4-2-6/h8-9H,3-7,10-11H2,1-2H3;5-6H,1-4H23
- 危险品标志:
-
T:Toxic
N:Dangerous for the environment
- 分子结构式:
-
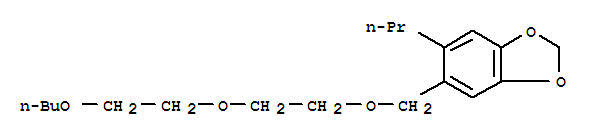
- SDS:
- 查看