硫雄甾醇
- 中文名称:
- 硫雄甾醇
- 中文别名:
- 硫雄甾醇;
环硫雄醇
- 英文名称:
- Epitiostanol
- 英文别名:
- Epitiostanol;
Epithiostanol;
Thiodrol;
Epithioandrostanol
- CAS No.:
- 2363-58-8
- 分 子 式:
-
C19H30OS
- 分 子 量:
- 306.5059
- 精确分子量:
- 306.20200
- PSA:
- 45.53000
- InChI:
- InChI=1/C19H30OS/c1-18-8-7-14-12(13(18)5-6-17(18)20)4-3-11-9-15-16(21-15)10-19(11,14)2/h11-17,20H,3-10H2,1-2H3/t11-,12-,13-,14-,15-,16+,17-,18-,19-/m0/s13
- 危险品标志:
-
- 风险术语:
-
- 安全术语:
-
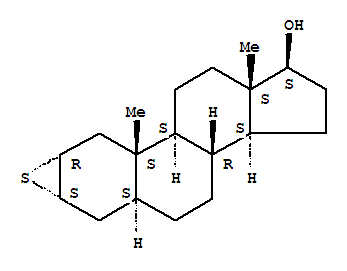
- 分子结构式:
-
- SDS:
- 查看
硫雄甾醇 物化性质
- 密度:
- 1.137 g/cm3
- 沸点:
- 424.4ºC at 760 mmHg
- 闪点:
- 206ºC
- 折射率:
- 1.579
- 蒸汽压:
- 5.56E-09mmHg at 25°C
硫雄甾醇 毒理性
CHEMICAL IDENTIFICATION
- RTECS NUMBER :
- KC1035000
- CHEMICAL NAME :
- 2-alpha,3-alpha-Epithio-5-alpha-androstan-17-beta-ol
- CAS REGISTRY NUMBER :
- 2363-58-8
- LAST UPDATED :
- 199706
- DATA ITEMS CITED :
- 18
- MOLECULAR FORMULA :
- C19-H30-O-S
- MOLECULAR WEIGHT :
- 306.55
HEALTH HAZARD DATA
ACUTE TOXICITY DATA
- TYPE OF TEST :
- LD - Lethal dose
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- >6 gm/kg
- TOXIC EFFECTS :
- Behavioral - somnolence (general depressed activity)
Liver - other changes
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 7,805,1973
- TYPE OF TEST :
- LDLo - Lowest published lethal dose
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 5 gm/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 7,805,1973
- TYPE OF TEST :
- LD - Lethal dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- >5 gm/kg
- TOXIC EFFECTS :
- Liver - other changes
Endocrine - other changes
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 7,805,1973
- TYPE OF TEST :
- LD - Lethal dose
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- >10 gm/kg
- TOXIC EFFECTS :
- Behavioral - somnolence (general depressed activity)
Lungs, Thorax, or Respiration - respiratory depression
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 7,805,1973
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 1160 mg/kg
- TOXIC EFFECTS :
- Behavioral - somnolence (general depressed activity)
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 7,805,1973
- TYPE OF TEST :
- LD - Lethal dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- >10 gm/kg
- TOXIC EFFECTS :
- Behavioral - somnolence (general depressed activity)
Endocrine - other changes
Skin and Appendages - hair
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 7,805,1973
- TYPE OF TEST :
- LD - Lethal dose
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Mammal - dog
- DOSE/DURATION :
- >150 mg/kg
- TOXIC EFFECTS :
- Behavioral - somnolence (general depressed activity)
Gastrointestinal - hypermotility, diarrhea
Gastrointestinal - nausea or vomiting
- REFERENCE :
- KSRNAM Kiso to Rinsho. Clinical Report. (Yubunsha Co., Ltd., 1-5, Kanda
Suda-Cho, Chiyoda-ku, KS Bldg., Tokyo 101, Japan) V.1- 1960-
Volume(issue)/page/year: 7,2983,1973
** OTHER MULTIPLE DOSE TOXICITY DATA **
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 3900 mg/kg/26W-I
- TOXIC EFFECTS :
- Liver - changes in liver weight
Kidney, Ureter, Bladder - changes in bladder weight
Endocrine - changes in spleen weight
- REFERENCE :
- KSRNAM Kiso to Rinsho. Clinical Report. (Yubunsha Co., Ltd., 1-5, Kanda
Suda-Cho, Chiyoda-ku, KS Bldg., Tokyo 101, Japan) V.1- 1960-
Volume(issue)/page/year: 7,2786,1973
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 8 gm/kg/40D-C
- TOXIC EFFECTS :
- Kidney, Ureter, Bladder - changes in bladder weight
Endocrine - other changes
Related to Chronic Data - changes in testicular weight
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 7,805,1973
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 1560 mg/kg/26W-I
- TOXIC EFFECTS :
- Kidney, Ureter, Bladder - changes in bladder weight
Endocrine - changes in thymus weight
Related to Chronic Data - changes in prostate weight
- REFERENCE :
- KSRNAM Kiso to Rinsho. Clinical Report. (Yubunsha Co., Ltd., 1-5, Kanda
Suda-Cho, Chiyoda-ku, KS Bldg., Tokyo 101, Japan) V.1- 1960-
Volume(issue)/page/year: 7,2786,1973
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 100 mg/kg/40D-C
- TOXIC EFFECTS :
- Kidney, Ureter, Bladder - changes in bladder weight
Endocrine - other changes
Related to Chronic Data - changes in testicular weight
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 7,805,1973
** REPRODUCTIVE DATA **
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intramuscular
- DOSE :
- 15 mg/kg
- SEX/DURATION :
- female 9-14 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - litter size (e.g. # fetuses per litter; measured
before birth)
Reproductive - Specific Developmental Abnormalities - musculoskeletal system
Reproductive - Specific Developmental Abnormalities - urogenital system
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 7,723,1973
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intramuscular
- DOSE :
- 15 mg/kg
- SEX/DURATION :
- female 9-14 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Newborn - live birth index (measured after birth)
Reproductive - Effects on Newborn - growth statistics (e.g.%, reduced weight
gain)
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 7,723,1973
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intramuscular
- DOSE :
- 60 mg/kg
- SEX/DURATION :
- female 9-14 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Newborn - weaning or lactation index (e.g., #
alive at weaning per # alive at day 4)
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 7,723,1973
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intramuscular
- DOSE :
- 12500 ug/kg
- SEX/DURATION :
- female 13-17 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death,
e.g., stunted fetus)
Reproductive - Specific Developmental Abnormalities - musculoskeletal system
Reproductive - Specific Developmental Abnormalities - urogenital system
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 7,723,1973
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intramuscular
- DOSE :
- 12500 ug/kg
- SEX/DURATION :
- female 13-17 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Newborn - growth statistics (e.g.%, reduced weight
gain)
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 7,723,1973
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intramuscular
- DOSE :
- 60 mg/kg
- SEX/DURATION :
- female 7-12 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Fertility - litter size (e.g. # fetuses per litter; measured
before birth)
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 7,723,1973
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intramuscular
- DOSE :
- 15 mg/kg
- SEX/DURATION :
- female 7-12 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Newborn - live birth index (measured after birth)
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 7,723,1973
推荐供应商
湖北恩星生物科技有限公司
硫雄甾醇
- 联系人:邵经理
- 电 话:16621771607
- 手 机:16621771607
- Cas号:2363-58-8
- 包装:100kg,200kg,300kg,400kg,500kg
- 纯度:
- 价格:64.0元 - 100.0元
湖北盛天恒创生物科技有限责任公司
环硫雄醇
- 联系人:陈经理
- 电 话:18971248750 18971247857 027-87676898 027-87734907
- 手 机:18971248750 18971247857
- Cas号:2363-58-8
- 包装:洽谈
- 纯度:
- 价格:洽谈

