返回
吲哚美辛法呢酯
- 中文名称:
- 吲哚美辛法呢酯
- 中文别名:
- 1-(4-氯苯甲酰基)-5-甲氧基-2-甲基-1-吲哚-3-乙酸3,7,11-三甲基-2,6,10-十二碳三烯酯;
吲哚美辛法呢酯
- 英文名称:
- indomethacin farnesil
- 英文别名:
- [(2E,6E)-3,7,11-trimethyldodeca-2,6,10-trienyl] 2-[1-(4-chlorobenzoyl)-5-methoxy-2-methylindol-3-yl]acetate;
3,7,11-Trimethyl-2,6,10-dodecatrienyl 1-(4-chlorobenzoyl)-5-methoxy-2-methyl-1H-indole-3-acetate;
1H-Indole-3-acetic acid,1-(4-chlorobenzoyl)-5-methoxy-2-methyl-,3,7,11-trimethyl-2,6,10-dodecatrienyl ester;
indometacin farnesil;
Infree (TN);
Infree;
E 0710;
Indomethacin farnesil
- CAS No.:
- 85801-02-1
- 分 子 式:
- C34H40ClNO4
- 分 子 量:
- 0
- 精确分子量:
- 561.26500
- PSA:
- 57.53000
- 分子结构式:
-
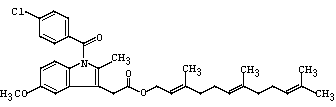
- SDS:
- 查看
推荐供应商
上海吉至生化科技有限公司
1-(4-氯苯甲酰基)-5-甲氧基-2-甲基-1-吲哚-3-乙酸3,7,11-三甲基-2,6,10-十二碳三烯酯
- 联系人:Shirley张
- 电 话:021-57520831
- 手 机:18116052675
- Cas号:85801-02-1
- 包装:100mgea
- 纯度:98%
- 价格:745.0元
湖北魏氏化学试剂股份有限公司
3,7,11-三甲基十二碳-2,6,10-三烯-1-基 2-(1-(4-氯苯甲酰基)-5-甲氧基-2-甲基-1H-吲哚-3-基)乙酸酯
- 联系人:刘涛
- 电 话:027-59101766
- 手 机:13125137661
- Cas号:85801-02-1
- 包装:100mg
- 纯度:98%
- 价格:776.0元
