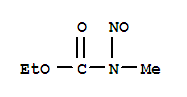返回
N-亚硝基-N-甲基尿烷
- 中文名称:
- N-亚硝基-N-甲基尿烷
- 中文别名:
- N-亚硝基-N-甲基尿烷;
N-甲基-N-亚硝基氨基甲酸乙酯
- 英文名称:
- Carbamic acid,N-methyl-N-nitroso-, ethyl ester
- 英文别名:
- ethyl N-methyl-N-nitrosocarbamate;
N-Nitroso-N-methyl-urethan;
N-nitroso-N-methylcarbamate;
Ethyl N-methylnitrosocarbamate;
N-nitroso-N-methylurethane;
Methyl-N-nitrosourethane;
NMUT;
methyl-nitroso-carbamic acid ethyl ester;
N-Nitro-N-methylurethan;
Methylnitrosourethane;
Methyl-nitroso-carbamidsaeure-aethylester;
N-Methyl-N-nitrosourethane;
Nitrosomethylurethane;
MNUN
- CAS No.:
- 615-53-2
- 分 子 式:
- C4H8N2O3
- 分 子 量:
- 132.12
- 精确分子量:
- 132.05300
- PSA:
- 58.97000
- EINECS:
- 210-432-3
- InChI:
- The Key: CAUBWLYZCDDYEF-UHFFFAOYSA-N3
- 分子结构式:
-
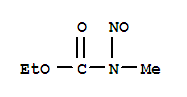
- SDS:
- 查看
推荐供应商
上海吉至生化科技有限公司
N-甲基-N-亚硝基氨基甲酸乙酯
- 联系人:Shirley张
- 电 话:021-57520831
- 手 机:18116052675
- Cas号:615-53-2
- 包装:1gea,5gea
- 纯度:96%
- 价格:57.0元 - 190.0元
宁波申泰生物科技有限公司
N-甲基-N-亚硝基氨基甲酸乙酯
- 联系人:高经理
- 电 话:0574-28802236
- 手 机:18606861312
- Cas号:615-53-2
- 包装:1kg
- 纯度:99%
- 价格:100.0元
武汉琼格生物科技有限公司
N-甲基-N-亚硝基尿烷
- 联系人:时燕
- 电 话:027-84235681
- 手 机:13657207535
- Cas号:615-53-2
- 包装:5g,5g,5g,5g,5g
- 纯度:0.95
- 价格:299.0元 - 299.0元